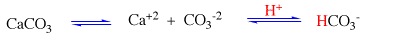DEAR AWESOMEVILLE...
You guys need to get a rain check! We cannot build this amazing statue under a cliff! Limestone is a calcium carbonate, which is the same as chalk! Acid rain is an acetic rain! When a calcium carbonate and an acetic acid mix, they bubble and sizzle as you saw in our experiments! The fizzing will make the cliff fall on top of the statue! We need to move the statue elsewhere before the cliff erodes and falls! Our hypothesis were sort of accurate by saying that it will fall and bubble. We were all off and acurrate on our hypthoesis. Blake thought that orange juice would be the most dangerous which didnt work out. Sarah was off thinking it would fall imediatly and Lauryn was off thinking it would be a bigger explosion. Other than that it was pretty acurrate. Acid rain is dangerous to the environment , lakes of water, and architecture. We should try to reduce use in cars and industrial factories.
I found out that when acidic acids and chalk ( calcium carbonate) mix, it forms a chemical reaction. We also found out the solute and solvents of the experiments and how it all works. The molecule for what happened is the above picture. The data supported our hypothesis that something would happen and carbon dioxide would form. The data showed that those acids and bases create a reaction. I learned how dangerous and how harmful acid rain is in bodies of water, forests, and architecture as stated in our introduction, which holds most of our information. I also learned how those acids and chalks react and to be more careful with ingredients. We also learned how when you add baking soda back into the chalk after being reacted on by vinegar, its properties come back. We mainly used a google doc in the information we researched. https://docs.google.com/a/alpineschools.net/document/d/16jhbj5e0I2S-o-8r36gIO_lBq46GqLZ-CRxZW4s3tmo/edit
I learned a lot about acetic acids and calcium carbonate and how they work.
One experimental error was that, the first time we combined chalk and vinegar and had to drain the vinegar, everything came out with it. When we tested it the second time, we waited a bit to let the chalk settle at the bottom before dumping the vinegar. Something that could have gone wrong is if we didn't add enough of an acidic base to the limestone, causing us to see a smaller, less visible reaction. Something else that could have gone wrong was if one of us were impatient and didn't wait for a reaction to occur. Some chemical reactions are not instantaneous, and require time to start reacting. If we wanted further research, we could have tested what other powders have similar pH's to limestone and baking soda, and see how they react to acid rain.
This experiment relates to real life, because acid rain DOES exist more on the eastern side of the United States, and in England. It occurs more in the east because of the prevalent emission of sulfur dioxide and nitrogen oxide, caused by industrial factories and other engines that require fuel. The acid from the fumes of factories are released into the air, carried by wind, and dissolve in the clouds' rainwater. After the clouds become heavy, they then precipitate like any rain cloud would. But instead of regular, neutral water, it consists of sulfur dioxide and nitrogen oxide. If acid rain precipitates on a fragile cliff or ledge, it could cause the ledge to slowly erode over time and to become smaller and smaller. It may even collapse as the acidic rain eats away at it, just like it did in Awesomeville. Acid rain also has the potential of killing wildlife, polluting water, and eroding stone statues. This is also an example of why it is unsafe to put a statue of Mr. Casey under a limestone cliff. Not only would the cliff collapse, but the rain could also eat away at his statue unless it is made of a different material than stone or something similar to that. We suggest placing his statue where it could stay dry.
~LOVE
LAURYN BLOTTIN, SARAH HARDY, AND BLAKE JEFFREYS (who's not batman)
I found out that when acidic acids and chalk ( calcium carbonate) mix, it forms a chemical reaction. We also found out the solute and solvents of the experiments and how it all works. The molecule for what happened is the above picture. The data supported our hypothesis that something would happen and carbon dioxide would form. The data showed that those acids and bases create a reaction. I learned how dangerous and how harmful acid rain is in bodies of water, forests, and architecture as stated in our introduction, which holds most of our information. I also learned how those acids and chalks react and to be more careful with ingredients. We also learned how when you add baking soda back into the chalk after being reacted on by vinegar, its properties come back. We mainly used a google doc in the information we researched. https://docs.google.com/a/alpineschools.net/document/d/16jhbj5e0I2S-o-8r36gIO_lBq46GqLZ-CRxZW4s3tmo/edit
I learned a lot about acetic acids and calcium carbonate and how they work.
One experimental error was that, the first time we combined chalk and vinegar and had to drain the vinegar, everything came out with it. When we tested it the second time, we waited a bit to let the chalk settle at the bottom before dumping the vinegar. Something that could have gone wrong is if we didn't add enough of an acidic base to the limestone, causing us to see a smaller, less visible reaction. Something else that could have gone wrong was if one of us were impatient and didn't wait for a reaction to occur. Some chemical reactions are not instantaneous, and require time to start reacting. If we wanted further research, we could have tested what other powders have similar pH's to limestone and baking soda, and see how they react to acid rain.
This experiment relates to real life, because acid rain DOES exist more on the eastern side of the United States, and in England. It occurs more in the east because of the prevalent emission of sulfur dioxide and nitrogen oxide, caused by industrial factories and other engines that require fuel. The acid from the fumes of factories are released into the air, carried by wind, and dissolve in the clouds' rainwater. After the clouds become heavy, they then precipitate like any rain cloud would. But instead of regular, neutral water, it consists of sulfur dioxide and nitrogen oxide. If acid rain precipitates on a fragile cliff or ledge, it could cause the ledge to slowly erode over time and to become smaller and smaller. It may even collapse as the acidic rain eats away at it, just like it did in Awesomeville. Acid rain also has the potential of killing wildlife, polluting water, and eroding stone statues. This is also an example of why it is unsafe to put a statue of Mr. Casey under a limestone cliff. Not only would the cliff collapse, but the rain could also eat away at his statue unless it is made of a different material than stone or something similar to that. We suggest placing his statue where it could stay dry.
~LOVE
LAURYN BLOTTIN, SARAH HARDY, AND BLAKE JEFFREYS (who's not batman)